Reltio Data Cloud®
Trusted system of context for business and AI.
Reltio Data Cloud® unifies, cleanses, and activates your data in real time, so you can achieve frictionless operations, real-time context intelligence, and agentic AI transformation.
Built for the operational reality of agentic AI.
Our scalable context intelligence platform fuels operational and analytical workflows with secure, high-quality data. Delivers fast time to value, agility, and genAI-powered innovation.
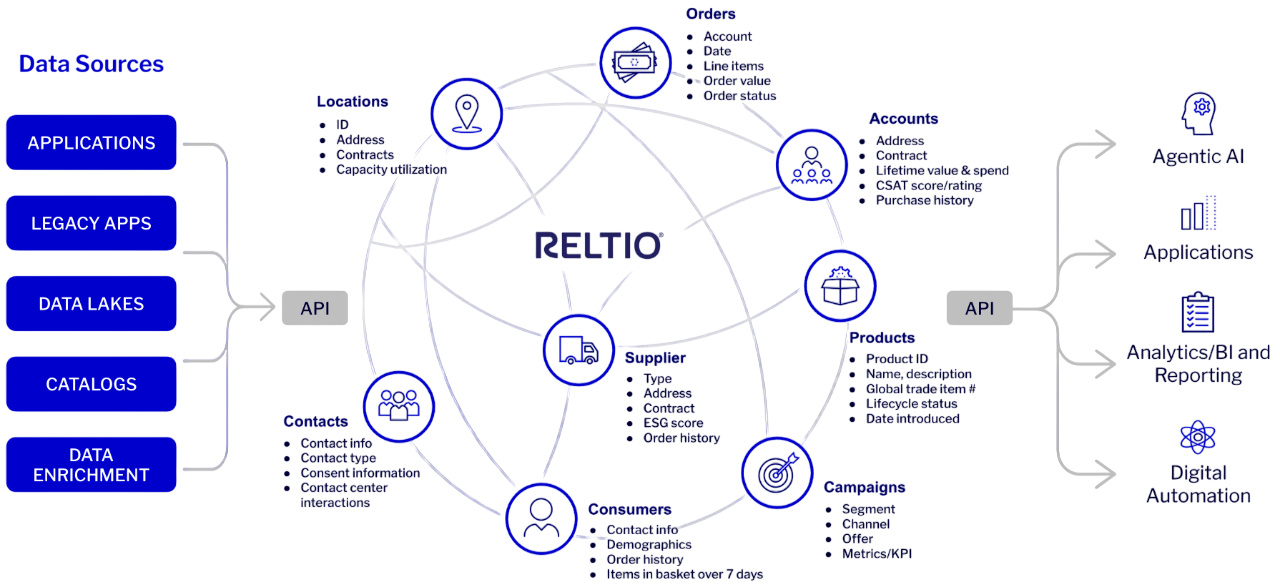
Relationship intelligence via data graph.
Our platform transforms siloed, multidomain data into a unified, graph-based unique profiles with real-time interactions and relationships.
Connected, organized data fuels modern digital experiences and agentic transformation. Your operational systems and AI agents have the structured context to understand enterprise data, make informed decisions, and perform complex tasks autonomously.

High-quality data for confident decisions.
LLM-powered FERN augments rule-based matching—boosting match accuracy and steward productivity. Built-in dynamic survivorship enables contextual views for different departments.
With continuous data profiling, quality validation, and reference data management, your teams always work with accurate, up-to-date data. Third-party enrichment improves match accuracy and adds critical context.
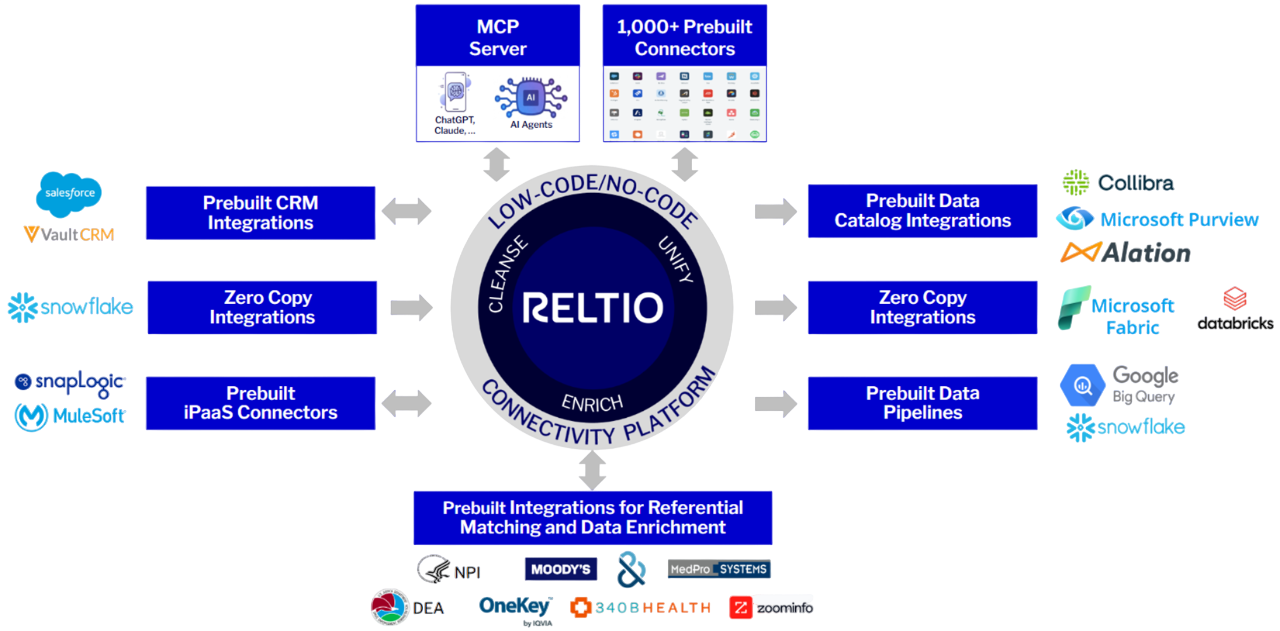
Fast, flexible data connectivity.
Our API-first platform provides over 1,000 prebuilt connectors in a low-code/no-code environment for real-time or batch integration needs. We offer zero-copy integration with major vendors to minimize the need to move the data.
Reltio Lightspeed™ Data Delivery Network enables access to trusted data with ≤50 ms latency to power your global, real-time applications and personalized experiences at touchpoint.

Built-in governance and trust.
Effectively govern your core business data with fine-grained security policies, data lineage, consent management, and audit logging. We help simplify compliance with HIPAA, GDPR, and other privacy regulations.
Our prebuilt integrations with leading data catalogs, such as Alation, Collibra, and Microsoft Purview, to share metadata streamline overall governance processes and build stakeholder confidence.

Time to value in < 90 days.
Purpose-built AI agents for data governance workflows, combined with industry- or domain-specific preconfigured components reduce your time to value to just weeks.
Covering key industries—financial services, insurance, life sciences, healthcare—our prebuilt canonical data models, integrations, and preset configurations bring the best practices used by industry leaders to your solution.
The foundation for all our products.
A secure, cloud-native SaaS platform that delivers agility and turns data into a competitive advantage in the age of AI.
Reltio Intelligent 360™
Reltio Multidomain MDM
Reltio AgentFlow™
Hear from our customers.





“We were promising three things: we were going to get better performance, the environment was going to become more stable. And we’re going to be spending about 80% of our time talking about innovation and 20% of our time doing the operations work. So that was what we had promised. And the nice thing is that it has essentially come to fruition.”
Chad McCord
Senior IT Manager, Master Data Management
“Reltio is at the heart of our digital innovation and customer experience initiatives. Reltio empowers 1,000s of sales reps and 100s of data stewards with the most up-to-date, trusted customer profiles… It allows us to manage customer data in a way that is agile, scalable, and smart.”
Joanna Walker
Global MDM Architect
“The prebuilt capabilities of the Reltio platform started with the heart of the solution—matching and merging. As a financial institution, we must be very careful with our members’ data, and Reltio had excellent security capabilities.”
Jay Franklin
Former Senior Vice President of Enterprise Data and Analytics
Why data warehouses and data lakes need Reltio for trusted AI.
By combining the scalability of cloud data warehouses and data lakes with the trusted context and agility of Reltio Data Cloud, organizations gain greater operational and analytical value from their existing data warehouse/lake investments.
Reltio complements data warehouses and data lakes by continuously delivering high-quality, governed, and context-rich data—enabling them to operate at full analytical speed and scale without being burdened by data quality or governance gaps.
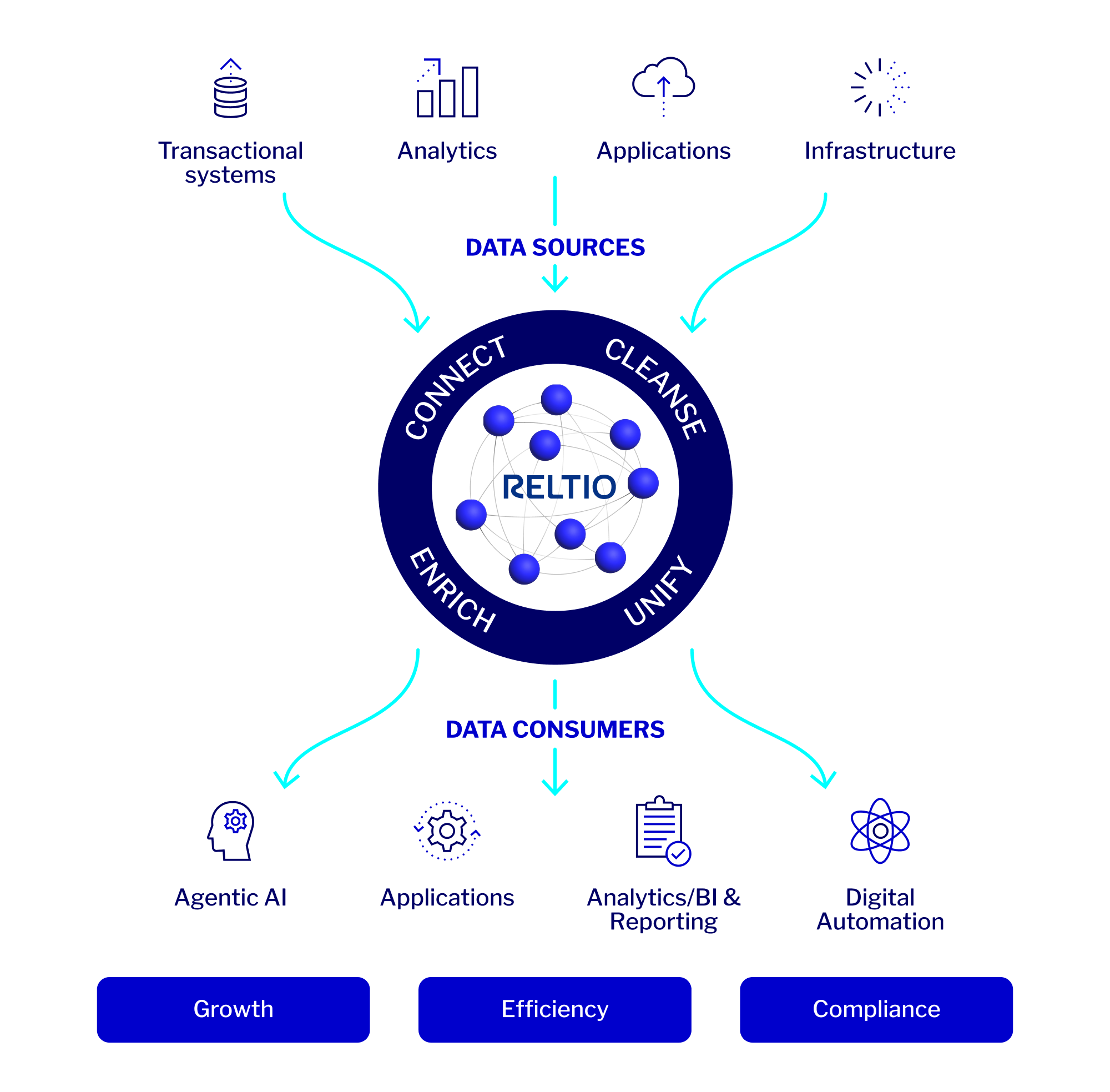
What makes Reltio Data Cloud different?
Our cloud-native SaaS platform operates with real-time performance, zero‑downtime upgrades, and enterprise-grade scalability. Designed to grow with your business, it keeps you ahead of change.
How it works.






Tenant provisioning.
Your secure SaaS instance is spun up with built-in security and high availability—ready to scale from day one.
Velocity pack load (Day 1).
Preconfigured industry data model, UI, match and other rules are automatically installed to give you an instant head start.
Source connection.
Prebuilt connectors and no-code integration platform ease data flow, without the delays of manual integration development.
Configure and iterate.
Fine-tune match, survivorship, and other rules. Review data quality dashboards, and run initial merges to tailor data to your needs.
Validation and consumption.
Data stewards validate records and business users interact with the data via UI and APIs getting the most from the platform.
Go-live (≤ 90 days).
First use case goes into production—often in under 90 days—followed by expansion into additional domains and use cases.
Fueling transformation and growth for every industry.
Ready to
see it in action?
See how the Reltio Context Intelligence Platform can give you the speed and flexibility you need to accelerate the value of your data and maximize its impact every day.







