Business Leaders
Transform with AI. Win with context intelligence.
Reltio turns siloed, complex data into trusted, actionable context intelligence that powers agentic AI at enterprise scale.

16 of the Fortune 100 and 43 of the Fortune 500 run on Reltio.
The age of intelligence demands a fundamental shift in how businesses operate.
AI is moving into core business operations—and without relevant, up-to-date business context, AI agents are fast, confident, and often wrong. The result? Stalled adoption and outcomes that fall far short of expectations.
From data chaos to business clarity.
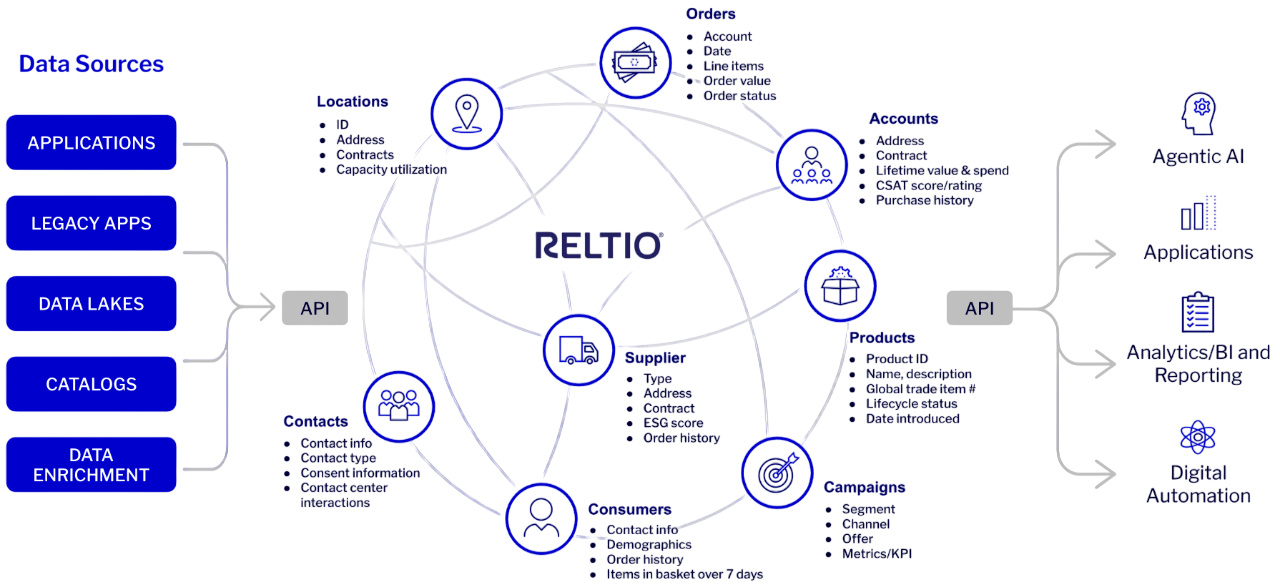
Reltio Context Intelligence Platform™ unifies data from all your systems into one governed, connected foundation that fuels agility and innovation. It delivers context intelligence—giving business leaders the confidence and speed needed to lead in the era of agentic AI.
Reltio Intelligent Data Graph™ brings that unified data to life by mapping relationships between customer, product, and supplier data—enriching it with insights from unstructured data, and turning complex connections into actionable intelligence.
Operate with clarity, confidence, and speed.
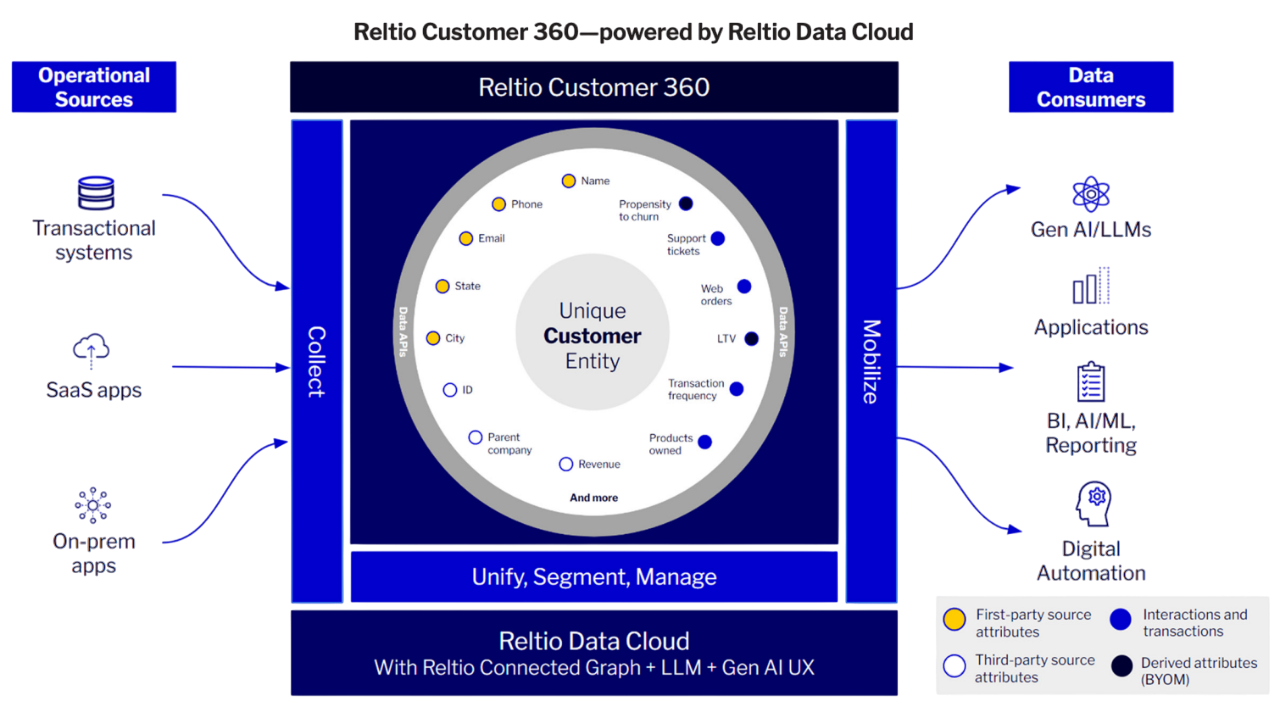
Alignment across the business starts with unifying data from disparate systems and formats into an up-to-date, governed, trusted view of the business. With rich context delivered in real-time, leaders make decisions faster and execute with confidence.
Boost your team's impact by delegating to AI agents with confidence.
AI systems need the same understanding of your business that your best people have—through trusted, rich data, connections, and meaning—so every agent can act with human-level judgement at enterprise scale.
Operate with agility and efficiency.
Boost real-time customer engagement with trusted, context-rich profiles delivered in milliseconds to any app or channel. So you can launch new experiences faster, engage with precision, and deliver seamless interactions that accelerate profitable growth.

Power better, faster decisions with richer context.
We harmonize, unify, and cleanse data across every source to deliver a single, trusted foundation for analytics and AI. With real-time visibility, sharper predictions, and faster time to insight, you can act with confidence.
Stay compliant, reduce risk, and lead with assurance.
With Reltio, governance and security are built in. Continuous governance keeps data trusted and compliant with strict data privacy laws and security standards, helping you manage risk and stay ahead of evolving regulations.

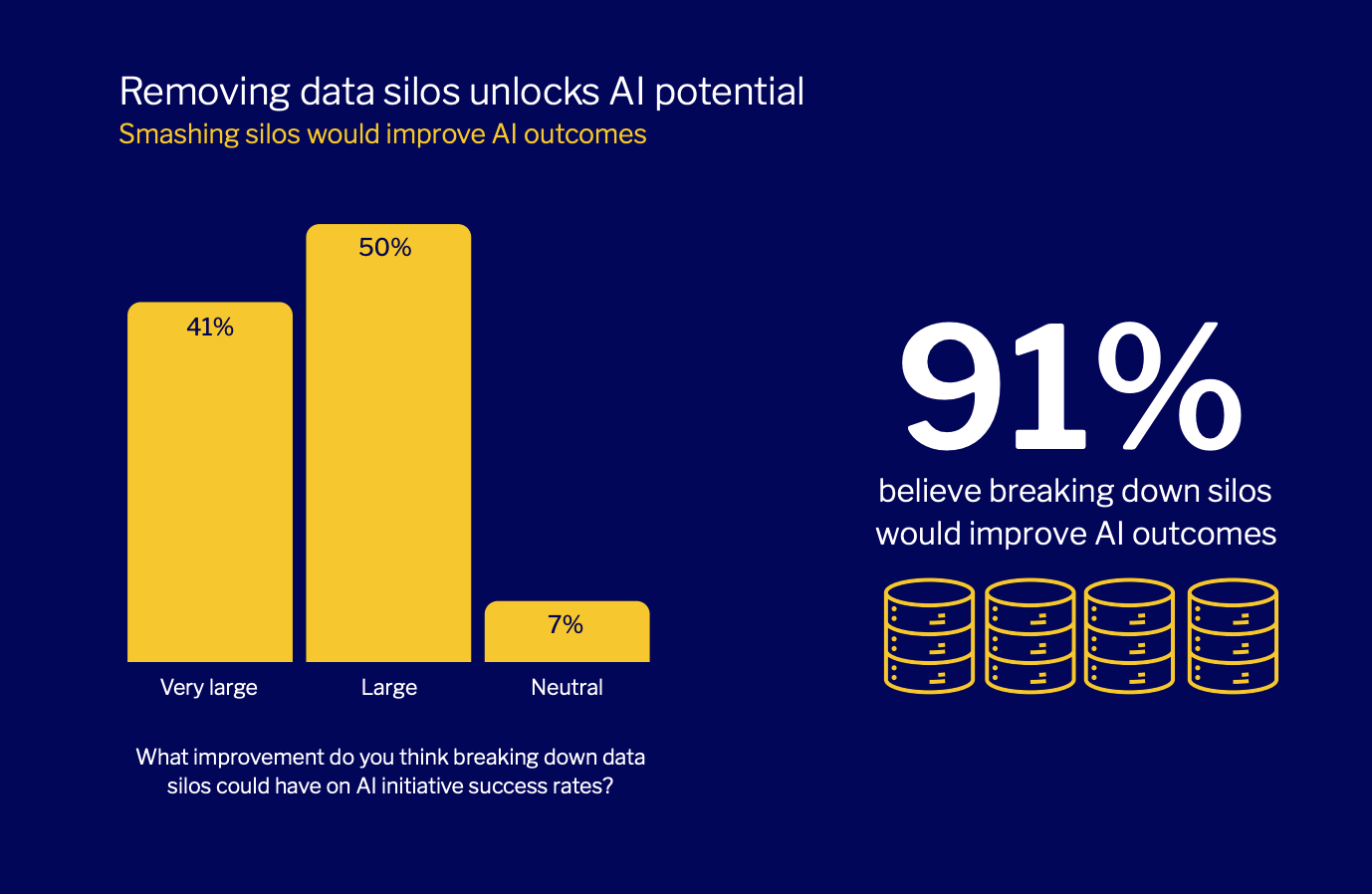
Harvard Business Review: Pulse Survey
Navigating Transformation in the Era of Agentic AI
Why business leaders choose Reltio.
We harmonize, unify, and cleanse siloed data into real-time business context for analytics, operations, and AI. Built for the operational reality of agentic AI, our platform gives you the clarity and control to power transformation with speed.

“Reltio’s core capabilities enable new customer experiences to quickly move from experiment to scale without creating additional dependencies.
We wanted a 360-degree view of our customers, and we couldn’t deliver that without having the data, systems, and technology together in a connected format.”
Shamim Mohammad
Executive Vice President & Chief Information and Technology Officer

Fueling transformation and growth for every industry.
Ready to
see it in action?
See how the Reltio Context Intelligence Platform can give you the speed and flexibility you need to accelerate the value of your data and maximize its impact every day.

























